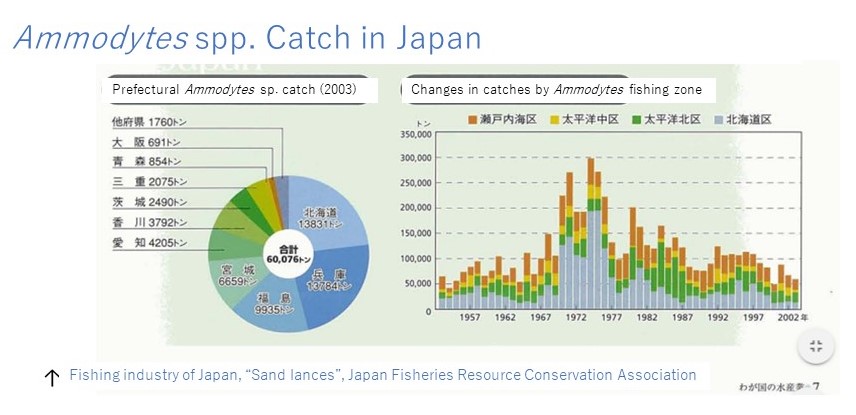
Life history of Ammodytes sp. in Mutsu Bay
セクションアウトライン
-
Generally:
· The stage observed immediately after hatching from an egg is known as larva. Many marine fish species, including Ammodytes spp., exhibit vulnerable morphologies that do not resemble the parent fish.
· As it subsequently grows, the cartilage transforms into the bone from the head to the tail (called “ossification”).
· In contrast, the fin rays are completed from the tail to the head in many fish, with the pectoral fins being complete at the end. When the numbers of fin rays in all fins are the same as those of the parent fish, the transformation is complete, and its name changes to that of a juvenile.
· Many larvae are planktonic (floating inhabitants).
These are the distinctions between early larval, late larval, and juvenile stages.



-
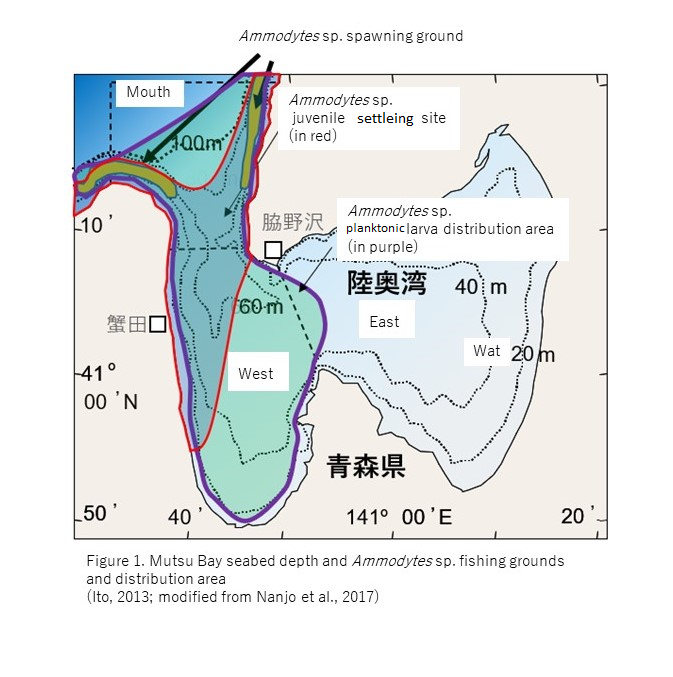
A spawning ground is observed for Ammodytes sp. on the seafloor at the mouth part of Mutsu Bay in Aomori Prefecture. The larvae that hatch on the seafloor around February and begin to live a plankton life in West Bay of Mutsu Bay.
There is unpublished data that two species of Ammodytes spp. are distributed in Mutsu Bay, but at the time of the following study, we recognized only one species (former Ammodytes personatus), so we refer to it here as Ammodytes sp.
When they subsequently grow into juveniles by May, they begin to live on the seafloor from the west side of the West Bay to the bottom of the mouth of the bay during the daytime (Fig. 1).
-
Ammodytes sp. matures at the age of 2-5 years and annually spawns 22,000-66,000 eggs per female. Of these eggs, if a total of two fishes (one male, one female) survive and mature to participate in spawning without dying of natural or fishing causes, then the Ammodytes resource will be maintained.
Based on the calculations from the number of spawned eggs, their survival rate is 0.0031 – 0.0089%, and the probability is as low as that of winning the lottery.
The survival rate is lower during the small and frail egg or larva/juvenile period than during the immature and adult fish stages. This is called “early reduction”. Furthermore, slight changes in environmental factors can have a large effect on early reduction. There are various theories as to why early reduction occurs. For example:
Starvation hypothesis: small larvae exhibit a slow swimming speed. Hence, low food densities can lead to hunger and death.
Transport hypothesis: Small larvae are plankton with poor swimming ability. Hence, they cannot swim to thermoclines (a layer of water in which its temperature and salinity change rapidly) and habitats that are easier to live in the way that nekton can. The survival rate would decrease if the larvae are carried away into the open ocean where they cannot survive due to abnormal ocean currents.
Water temperature hypothesis: Sudden drops in water temperature or high water temperatures result in mass death since they cannot escape.
-
Differences in the survival rate of 1% per day would result in a 40-fold difference after one year.
-
Should nurseries be released or be converted into aquaculture?
-
-
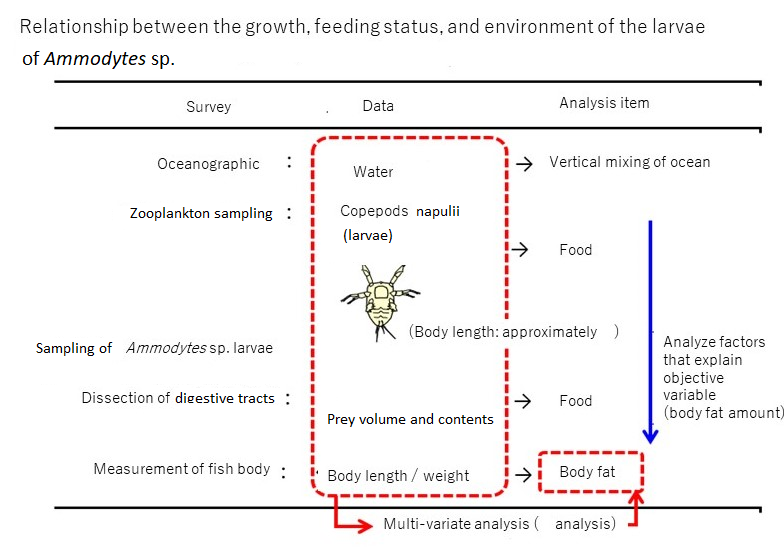
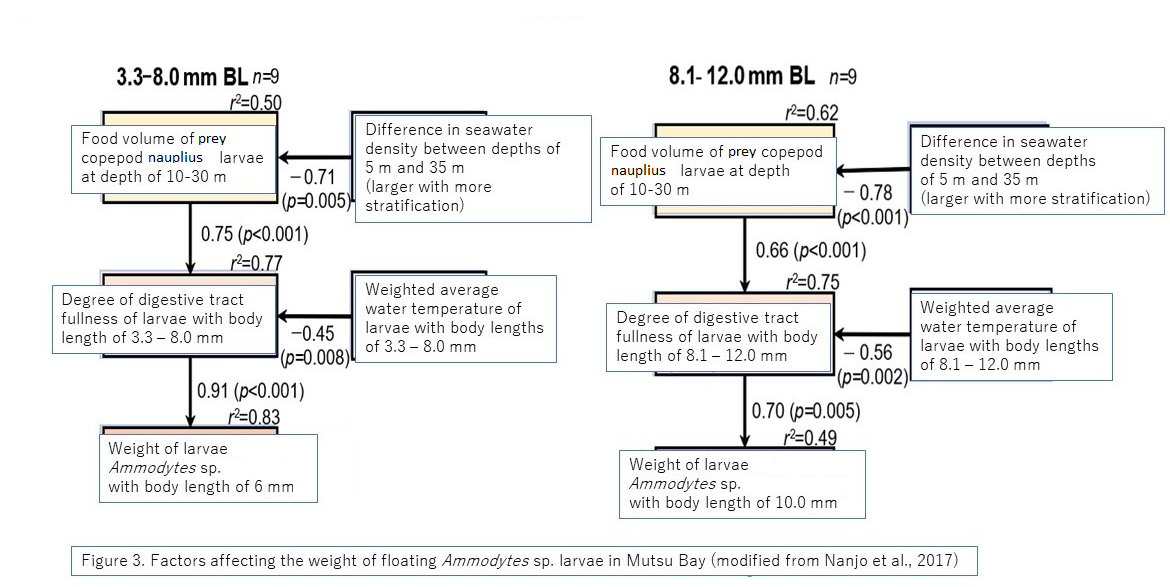
Path analysis results (Fig. 3)
Result 1: Separate analyses were conducted for early larvae (left) and late larvae (right). The values on the side and bottom of the arrows refer to the path coefficient β (a sort of directional correlation coefficient). Positive values showed a positive correlation, whereas negative values showed a negative correlation.
-
Relationship between growth, feeding status, and environment of Ammodytes sp. larvae (analysis/discussion)
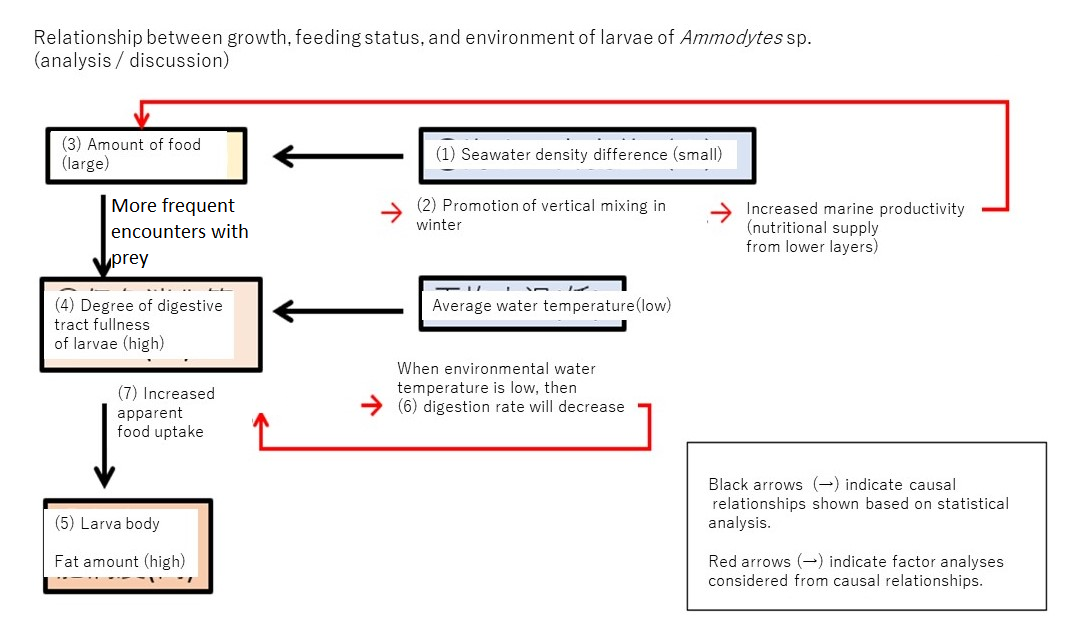
1. If the seawater density difference between depths of 5 m and 35 m is small (i.e., the inflow of river water into Mutsu Bay is small),
2. then vertical mixing in winter is promoted,
3. the number of food organisms increases,
4. more food is eaten,
5. and nutrients are accumulated between the periods of body lengths of 6 mm and 10 mm, and they become fat.
These could be proved statistically.
However, under low water temperatures:
6. the digestion rate slows down,
7. the food intake increases and the energy intake decreases,
but the effect of (3) (absolute values of β = 0.75 and 0.66) is greater than the effect of (6) (absolute values of β = −0.45 and −0.56[A1] ),
so it was statistically shown that the water temperature environment of Mutsu Bay was not as strong an effect for decreasing (5) body fat amount (can be calculated as an effect of 0.60 times (= 0.45 ÷ 0.75) to 0.85 times (= 0.56 ÷ 0.66).
-
Differences in the yearly survival rate of Ammodytes sp. in Mutsu Bay are shown.
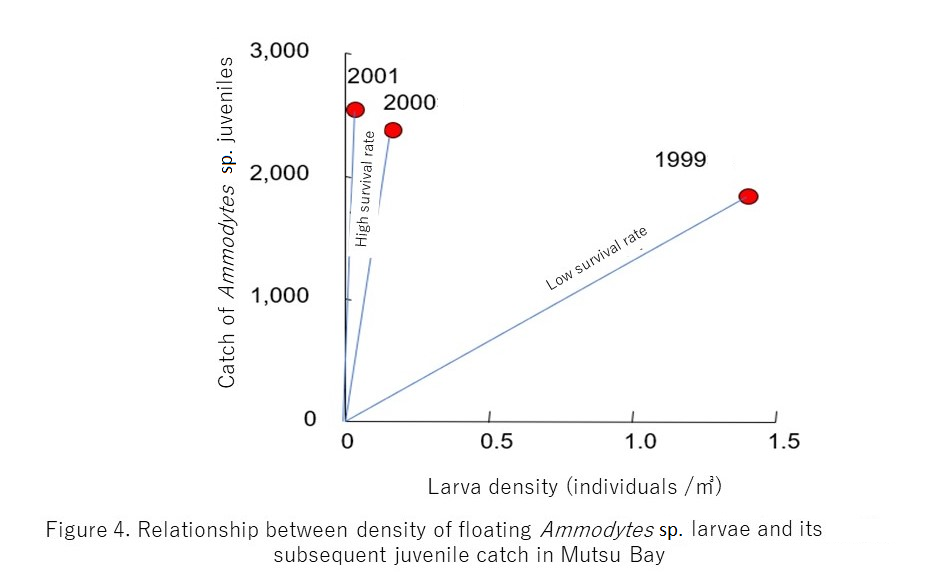
· The year 1999 demonstrated the highest density of larvae in three years but showed the fewest surviving juveniles. In other words, this signifies that there was large mortality between larval and juvenile periods. The slope of the line in the figure is small, so 1999 exhibited a low survival rate.
· In contrast, 2001 demonstrated a high survival rate.
-
-
Publicly available data can be used for analysis.
-
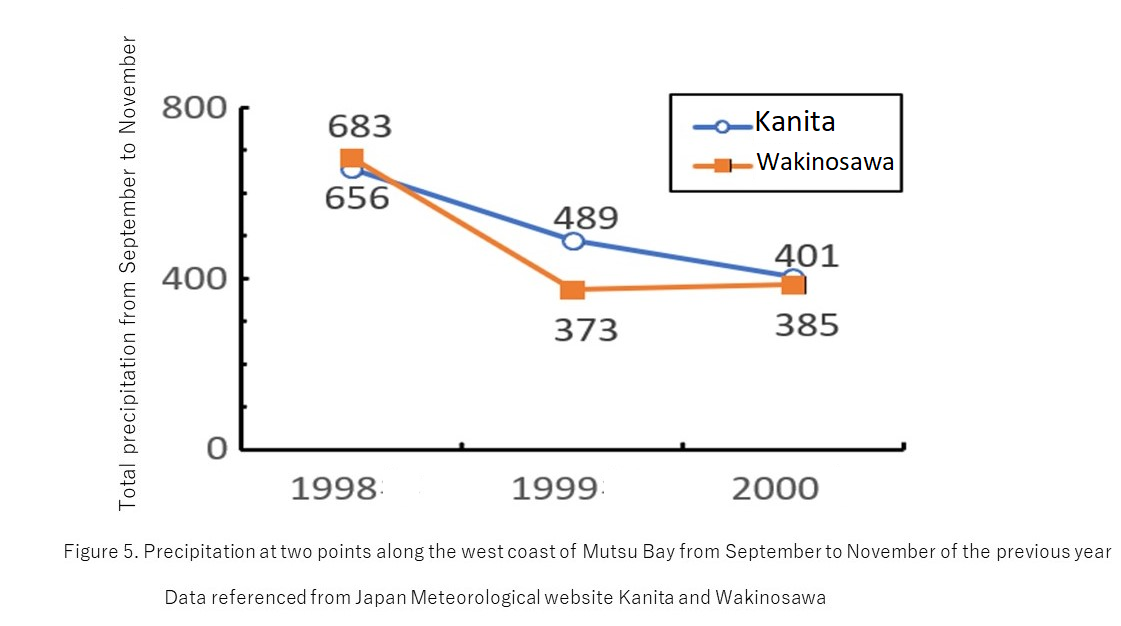
Discussion: combining results 1-3,
We can predict that during the years when a large amount of precipitation is observed in the fall, a decrease in the amount of Ammonytes sp. is observed in the spring.In these years, it may be necessary to control the catch in order to avoid overfishing.
How is the larva density, shown in Figure 4, determined?
The number of eggs is generally proportional to the body weight of the female parent. Hence, if the nutritional status of the female parent is the same every year, then the number of eggs can be determined by the number of parents.
However, the survival rate of larvae fluctuates greatly due to natural phenomena as examined here, and there is no room for human control.
The very least that humans can do (aside from seedling release) is to increase the number of female parent fishes by avoiding overfishing. Furthermore, prohibiting fishing would result in the growth of older and larger parent fish, and the number of eggs spawned would also increase. At that point, we would have to wait for years when the survival rate of larvae is high.