Search for health functional ingredients in red algae, a low-use resource; a method for preparing new oligosaccharides2
セクションアウトライン
-
In "Development of a New Oligosaccharide Preparation Method from Red Algae, a Low-Use Resource," we introduced a method for preparing xylooligosaccharides, which have a structure unique to dulse. The preparation method used commercially available enzymes, which include two types of enzymes: endo-type enzymes that hydrolyze the polysaccharides from the inside and exo-type enzymes that hydrolyze the polysaccharides from the ends. Exo-type enzymes turn oligosaccharides into the monosaccharide xylose, thus reducing oligosaccharide yields. In this course, we prepared only the endo-type enzyme by acquiring the gene for the endo-type enzyme from microorganisms and having E. coli produce the enzyme based on the genetic information. We used it to prepare oligosaccharides.
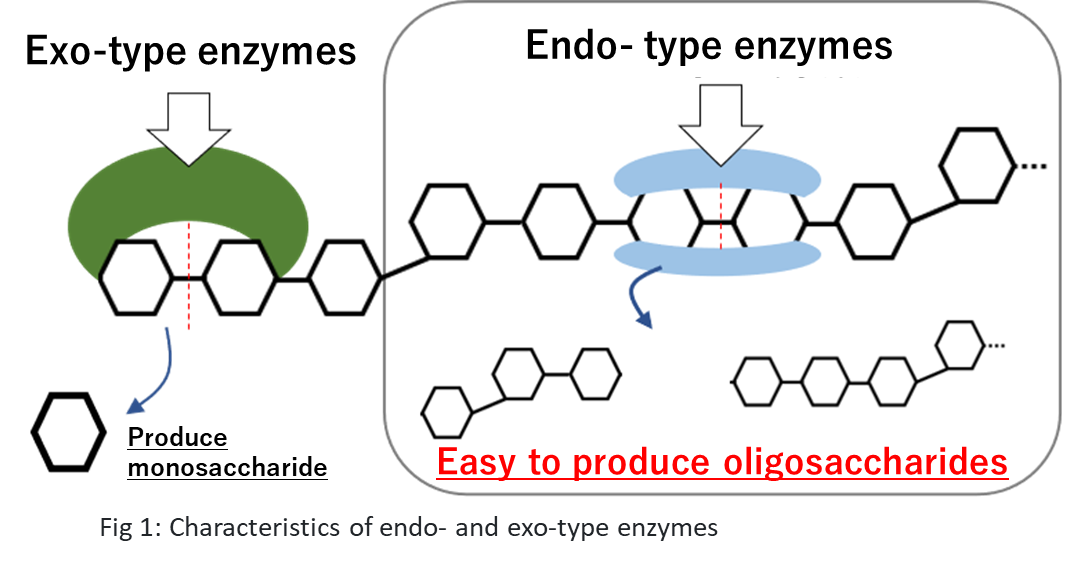
-
The actinomycete Streptomyces thermogriseus NBRC100772 grows in warm environments. Many of the enzymes of microorganisms living in such environments are heat stable. Stable enzymes are less likely to be deactivated, making them easier to study and use in industry. For this reason, we have prepared genomic DNA from this microorganism. This bacterium contains two xylanase genes, which were cloned, and an E. coli expression system was constructed to prepare the target enzyme. 2 xylanase gene information is registered in the DNA Data Bank of Japan (DDBJ) (StXyl10: LC603131; StXyl11: LC 603130).
-
The properties of the prepared enzymes were investigated. Both enzymes were stable at optimum temperatures above 60°C and in a wide range of pH optima (5.0-9.0). When placed at various temperatures for extended periods of time, StXyl10 was stable at 50°C, while StXyl11 showed a slow decrease in activity under temperature maintained at 60°C.
Figure 2: Basic properties of the two enzymes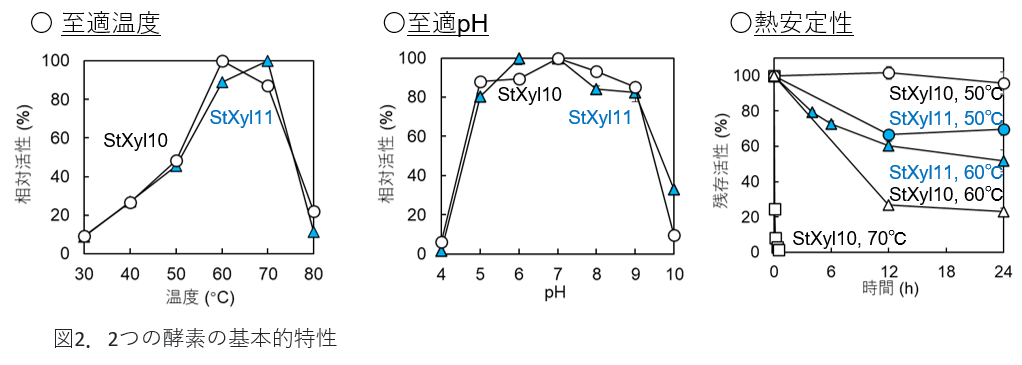
-
The amount of enzyme and reaction time were examined as conditions for degradation of xylan. After a certain reaction time, the reaction was subjected to thin-layer chromatography, and the concentration of the detected spots was used to determine what size oligosaccharides were being produced. We then determined the conditions under which the amount of monosaccharide produced was reduced and the amount of oligosaccharide produced was increased.
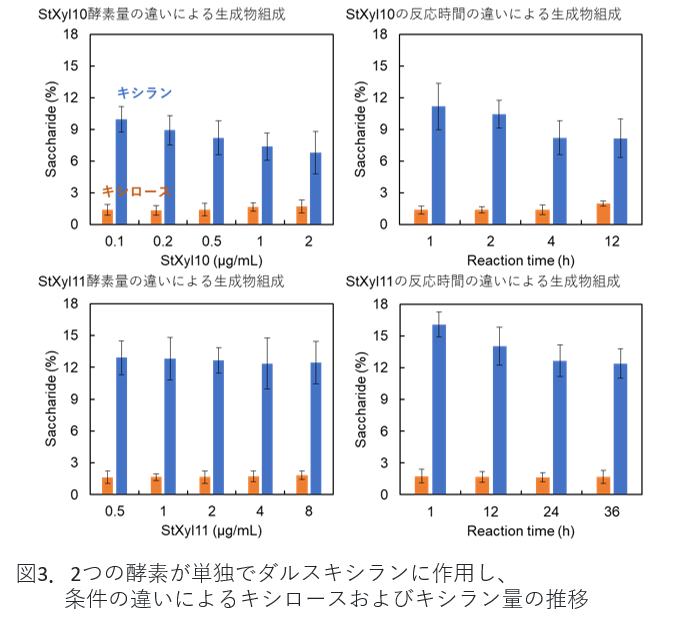
Figure 3. xylose and xylan amounts under different conditions with the two enzymes acting alone on dulse xylan. -
After examining the products using the above methods, we were finally able to combine the two enzymes to produce less xylose and more oligosaccharides. The products were subjected to gel filtration chromatography to confirm the long chain length carbohydrates and high performance liquid chromatography to analyze the short chain length oligosaccharides. The figure below summarizes these results. Oligosaccharides with β-(1→3)-glycosidic linkages (DXOS), which are characteristic of dulse, are shown in red, and those consisting only of β-(1→4)-glycosidic linkages are shown as β-(1→4)-XOS with blue X2-X4. Thus, we were able to prepare oligosaccharides containing a large amount of DXOS, which is characteristic of dulse. The amount of xylose at this time was less than 1%, indicating efficient preparation of oligosaccharides.
Note: DX or X followed by the number of xylose indicates the number of xylose. For example, X2 means disaccharide.
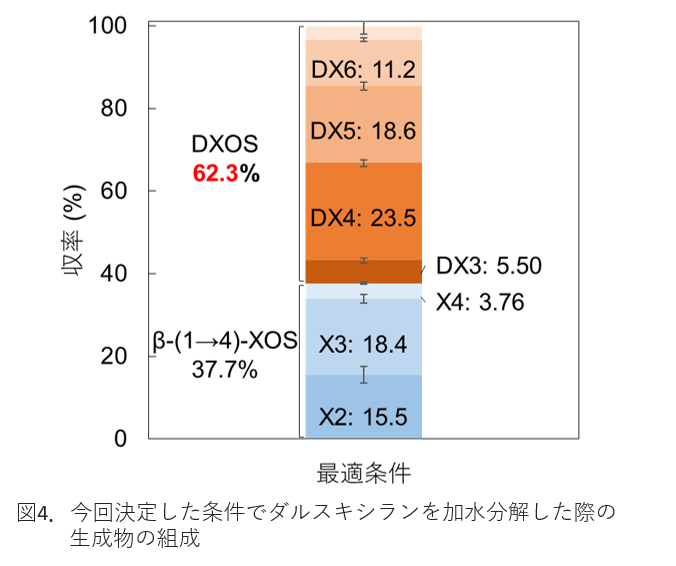
Figure 4: Composition of products obtained from the hydrolysis of dulse xylan under the conditions determined in this study.
-
Fujii et al. Preparation of β(1→3)/β(1→4) xylooligosaccharides from red alga dulse by two xylanases from Streptomyces thermogriseus
Open Access, so you can browse freely. -
・Development of preparation method of xylooligosaccharides from dulse with different structures
・Functional evaluation for beneficial bacteria
-




