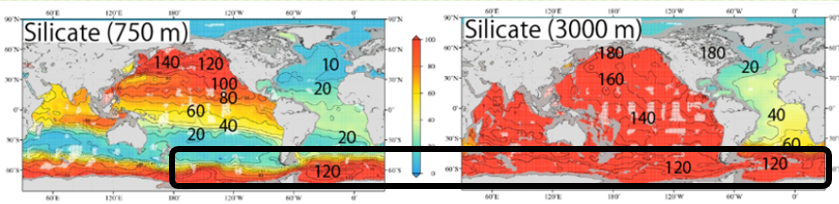
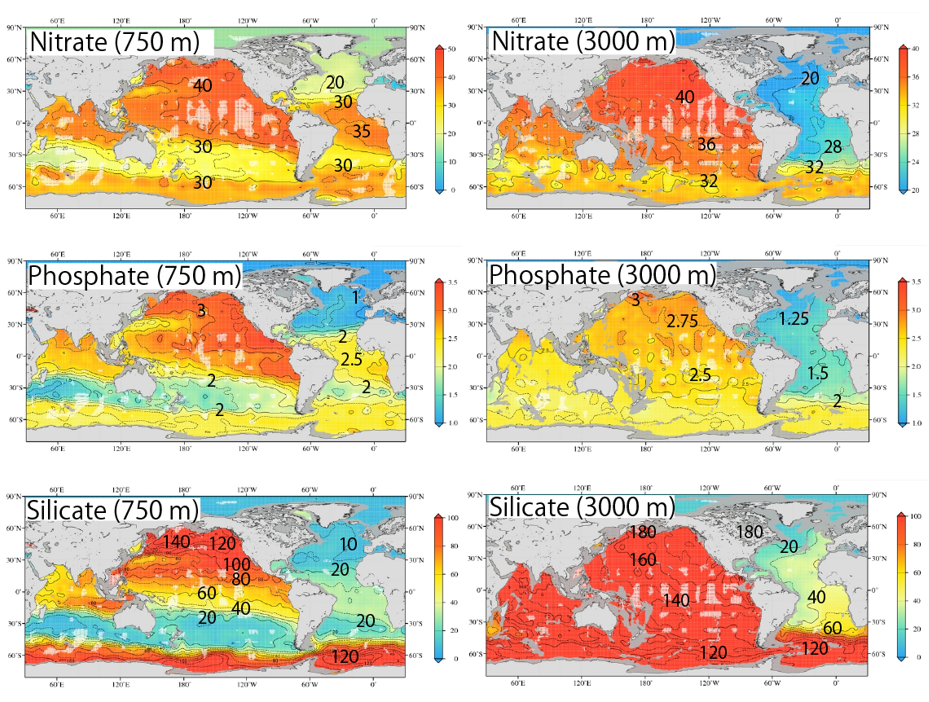
The global distribution of the three main nutrients in the pycnocline (750 m) and the deep layer (3000 m) are shown side by side. The pycnocline is on the left and the deep layer is on the right, from top to bottom: nitrate, phosphate, and silicate. I added the concentration values to the map so that the changes in concentration can be seen. Please compare the three components again.
As explained on the previous page,
Commonalities at a glance
① Each nutrient component is more concentrated in the deep North Pacific than in the deep North Atlantic (3000 m).
② For each nutrient component, the North Pacific and Antarctic Oceans have high concentrations at a depth of 750 m.
Differences that are not apparent to the ordinary person at a glance
③ Only silicic acid differs in concentration ratio between the deep North Atlantic and the deep North Pacific.
Nitrate and Phosphate:【Deep North Atlantic Concentration】×2=【Deep North Pacific】
Silicate :【Deep North Atlantic Concentration】×8=【Deep North Pacific】
(Silicic acid concentrations are significantly higher in the deep North Pacific than in the deep North Atlantic)
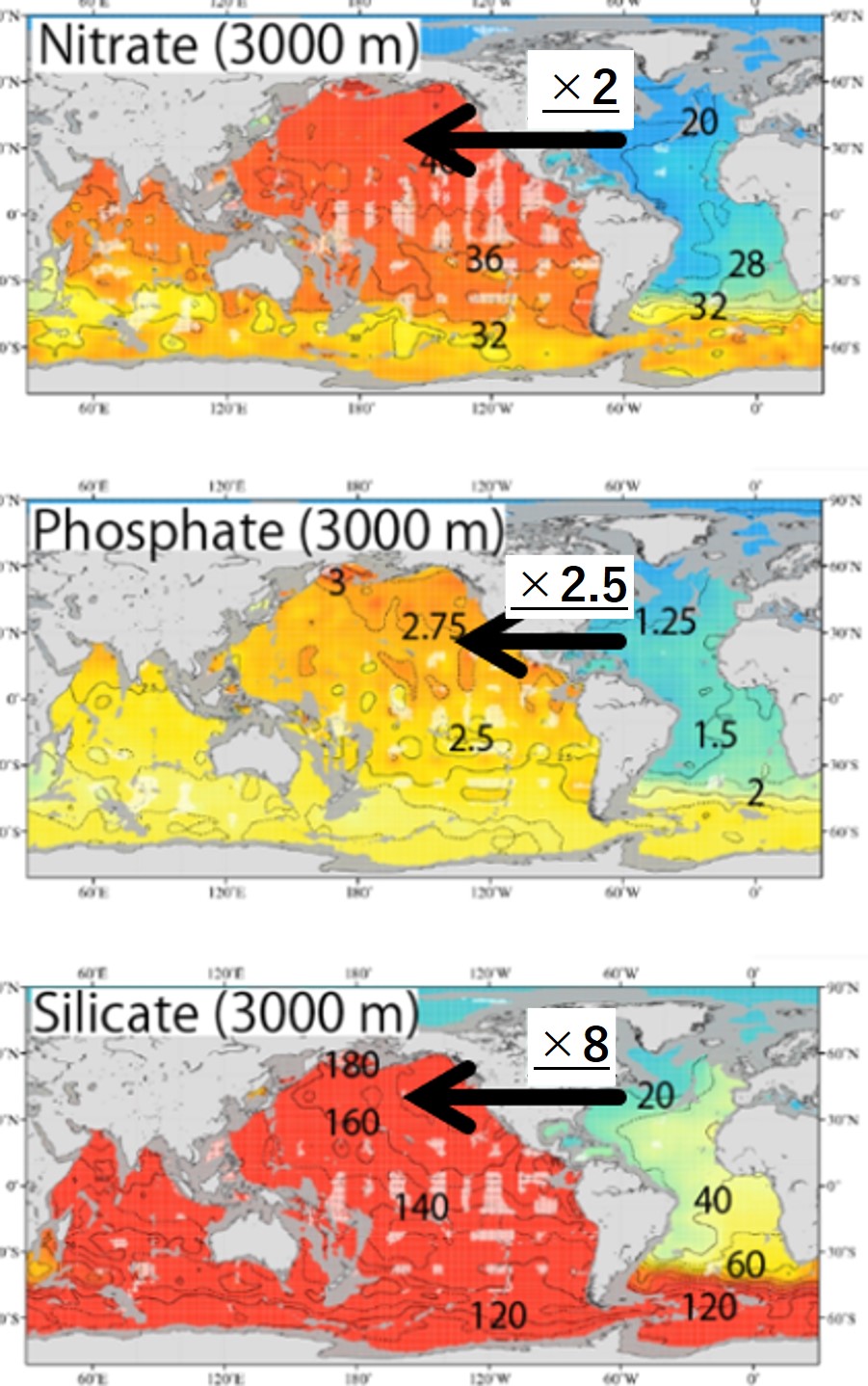
Characterizing these differences is the rather slow rate of dissolution of silicic acid (Si(OH)4) from silica shells (Opal: SiO2) in seawater; P and N are contained in the soft tissue of organic matter and are quickly decomposed and regenerated during the settling of particulate organic matter. Therefore, the concentrations of PO43- and NO3- increase rapidly in the pycnocline above the deep water. In contrast, silica shells (Opal) do not decompose much during settling. After being deposited in deep water or on the seafloor, silica shells (SiO2) gradually decompose and Si(OH)4 accumulates in the deep water. As a result, Si(OH)4 concentration maxima appear in the deep to bottom layers. The Si(OH)4 concentration in deep North Pacific water is considerably higher than that in deep North Atlantic water.
Here are the differences I found at a quick glance.
It is fun not only to look at the figure and memorize the characteristics described in the textbook, but also to find the characteristics and consider the causes on your own. From a quick glance at the features, it is a good idea to express them numerically. What does that number mean? It will broaden your consideration.
④ Difference in concentration between the deep layer and pycnocline (South Atlantic and South Pacific)
Nitrate and Phosphate:【Pycnocline】×1.25=【Deep layer】
Silicate :【Pycnocline】×2~5 =【Deep layer】
(Silicic acid concentration is more than twice as high in the deep layer as in the pycnocline.)
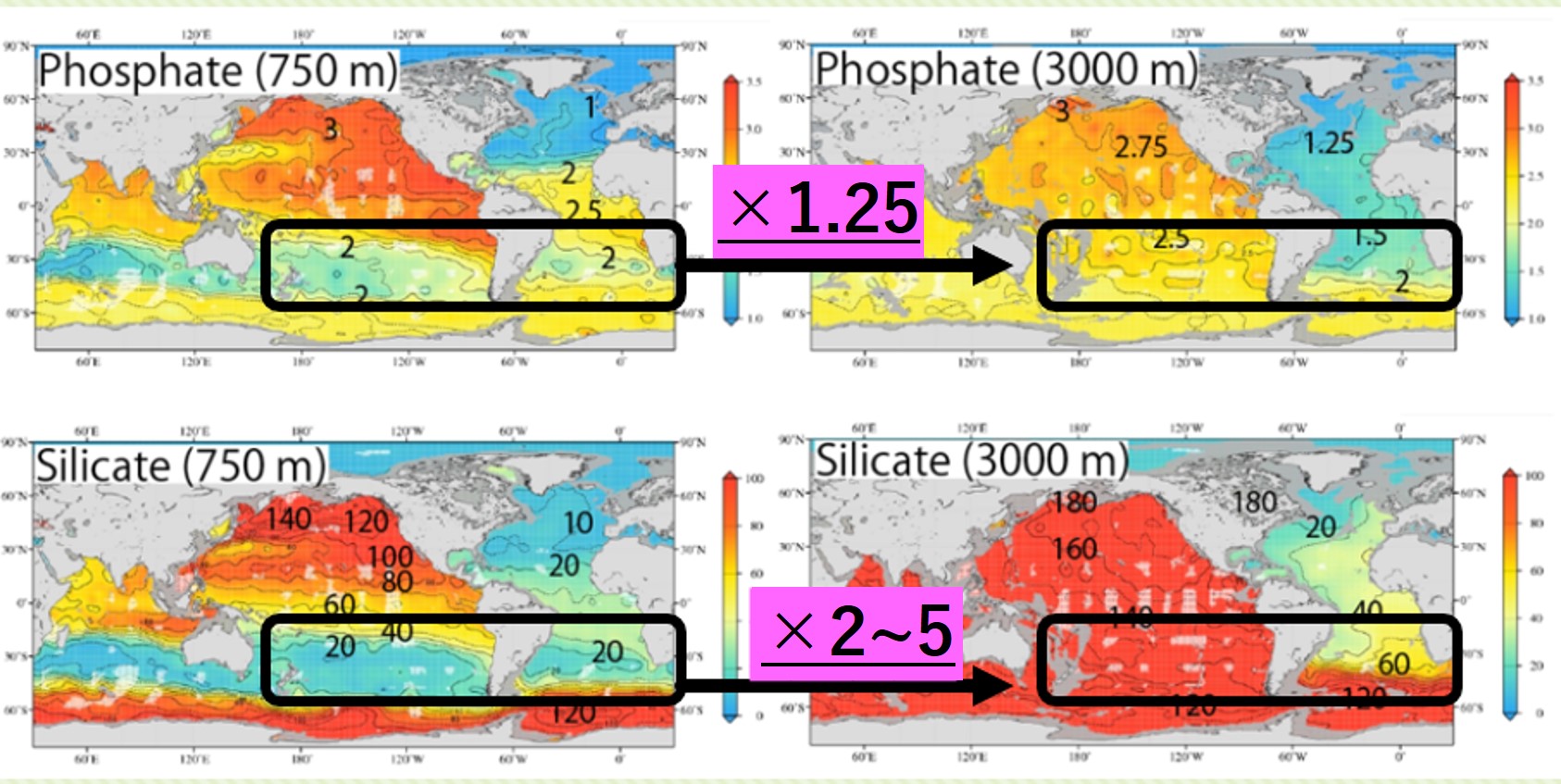
Discussion of reasons for the sudden increase in Si(OH)4 concentrations in the deep Atlantic to deep Southern Ocean
When entering the Southern Ocean from the Atlantic Ocean, Si(OH)4 concentrations increase rapidly in the deeper layers (from 60 to 120 μmol/L). This does not mean that the rate of decomposition of silicate shells (Opal) increases rapidly when Atlantic water enters the Southern Ocean. In the Southern Ocean, the Antarctic Circumpolar Current is circling around and considerable time has passed, so this is thought to be the result of the silicate shells accumulating Si(OH)4 as they gradually decompose. Therefore, we thought that the concentration of Si(OH)4 would suddenly increase at the confluence of the North Atlantic deep water and the Antarctic Circumpolar Current.
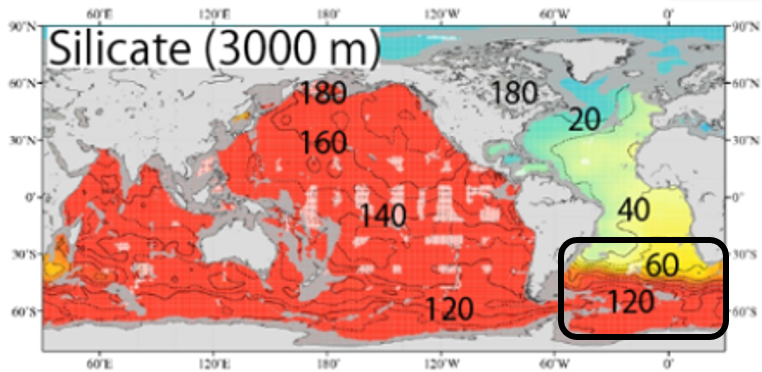
Note that in the Antarctic Ocean (Southern Ocean), Si(OH)
4 concentrations are as high at a depth of 750 m as at a depth of 3000 m. This is probably the result of intense vertical convection, which supplies Si(OH)
4 accumulated in the deeper layers to the 750 m.